Subtle Medical Receives FDA 510(k) Clearance for AI-Powered SubtleMR™
MENLO PARK, Calif., Oct. 15, 2019 /PRNewswire/ — Subtle Medical, a healthcare technology company focused on improving the quality, efficiency, and accessibility of medical imaging, announced today 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market SubtleMRTM. SubtleMR is an image processing software that uses denoising and resolution enhancement to improve image quality.
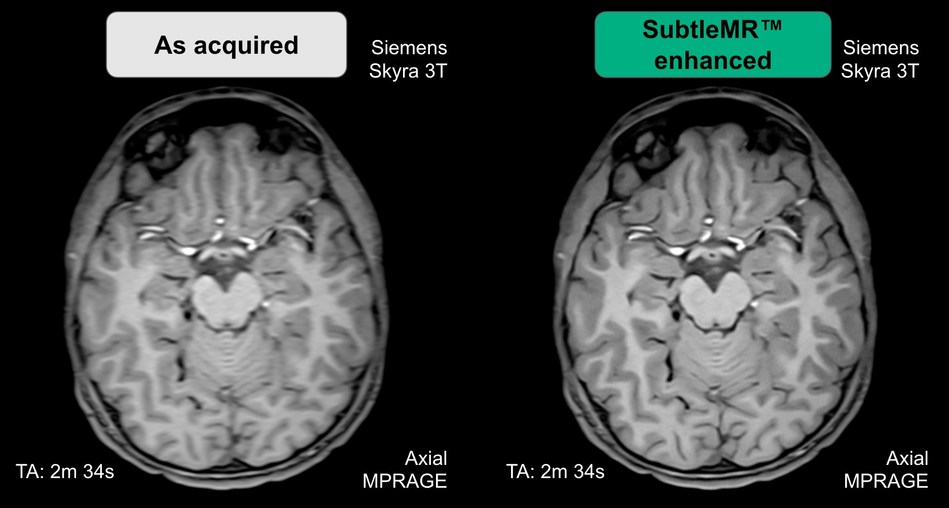
SubtleMR utilizes proprietary deep learning algorithms to bring the latest imaging enhancement technology to existing scanners. It is currently in pilot clinical use in multiple university hospitals and imaging centers.
“One of the most exciting things about deep learning reconstruction is how it redefines the usual negotiation between exam time and image quality. This could lead to significant downstream value for imaging operations and for patient experience,” said Christopher Hess, MD, Chair of the Department of Radiology and Biomedical Imaging at UCSF.
SubtleMR delivers a significant improvement in the quality of noisy images, which is particularly beneficial for patients who have difficulty holding still for long periods of time. Artifact-ridden images and the need for re-scans are a challenge for both patients and physicians. SubtleMR integrates seamlessly into the radiology workflow, and it is compatible with any brand of MRI scanner and PACS.
Subtle Medical’s focus is on applying the latest deep learning algorithms to image acquisition, an area of considerable importance in healthcare given the millions of scans conducted at busy radiology departments and imaging centers. “We are pleased to receive FDA clearance for SubtleMR, and we look forward to helping radiology departments and imaging centers get the most out of their existing MRI scanners,” said Enhao Gong, PhD, Founder and CEO of Subtle Medical. “This is an important milestone for the company as it broadens our portfolio of technologies developed for radiologists and their patients.”
Subtle Medical has the first and only AI solutions to receive FDA clearance for medical imaging enhancement—SubtlePET™and SubtleMR™. The company’s first product, SubtlePET, received FDA Clearance in 2018 for the enhancement of images from up to 4x faster PET scans. Subtle Medical’s third product in development, SubtleGADTM, has the potential to reduce gadolinium dosage during contrast-enhanced MRI exams. The company was recently awarded a $1.6 million NIH grant to further its research on this technology.
About Subtle Medical, Inc.
Subtle Medical, Inc. is a healthcare technology company founded by Stanford University Neuroradiologist, Professor Greg Zaharchuk, MD, PhD and Enhao Gong, PhD. The company has developed a suite of deep learning solutions that improve workflow efficiency and patient experience. It received the NVIDIA Inception Award for Top Healthcare+AI Startup Globally selected from over 3,000 contenders. It also was named a 2018 Minnies Award semi-finalist for Best New Radiology Vendor by AuntMinnie.com. Current clinical partners include UCSF, Hoag Hospital, UCSD, UPMC, MD Anderson and more. For more information, visit subtlemedical.com.
Contact: Anna Menyhart-Borroni
Email: anna@subtlemedical.com
Phone: 501-766-2662
SOURCE Subtle Medical, Inc.


