MedTech Innovator is the industry’s nonprofit global competition and accelerator for medical device, digital health and diagnostic companies. Our mission is to improve the lives of patients by accelerating the growth of companies that are transforming the healthcare system.
MedTech Innovator Unites Leading Players in the Medtech Innovation Ecosystem
Manufacturers
Our corporate partners include leading manufacturers like Johnson & Johnson, Becton Dickinson, Dexcom, Edwards Lifesciences, LivaNova, Olympus Corporation of the Americas, Samsung Research America, Zimmer Biomet, and many others. They participate as thought leaders, application reviewers, event judges, and accelerator mentors.
Conferences
In 2026, MedTech Innovator will hold events, competitions and showcases at JP Morgan Healthcare in San Francisco, the Wilson Sonsini Medical Device Conference in San Francisco; The MedTech Strategist in Dublin, Ireland; Device Talks in Boston, MA; AdvaMed’s The MedTech Conference; and ApacMed’s MedTech Forum in Singapore.
Providers
Providers like UCLA Health, Hackensack Meridien, Mayo Clinic, UnityPoint Health, Highmark Health, Providence, and Children’s National participate as application reviewers and judges and give critical feedback on topics like healthcare economics, customer requirements, integration issues, and more.
Investors
Hundreds of investors like RCT Ventures, Aphelion Capital, Life Science Angels, Healthtech Capital, Sofinnova Partners, Vensana, Treo, Hunniwell Lake Partners, Seroba Life Sciences participate as application reviewers, mentors, and judges
Trade Associations
MedTech Innovator has partnered with AdvaMed and AdvaMed Accel in the US and APACMed in Asia to provide best-in-class start-ups access to a virtual accelerator and to support members developing innovative and transformational medical technologies.
Service Providers
Experts provide critical feedback to MedTech Innovator companies from leading service providers like Asahi Intecc, Wilson Sonsini, Medmarc, Syensqo, NAMSA, and Proxima Clinical Research.
The Largest Accelerator of Medical Technology in the World
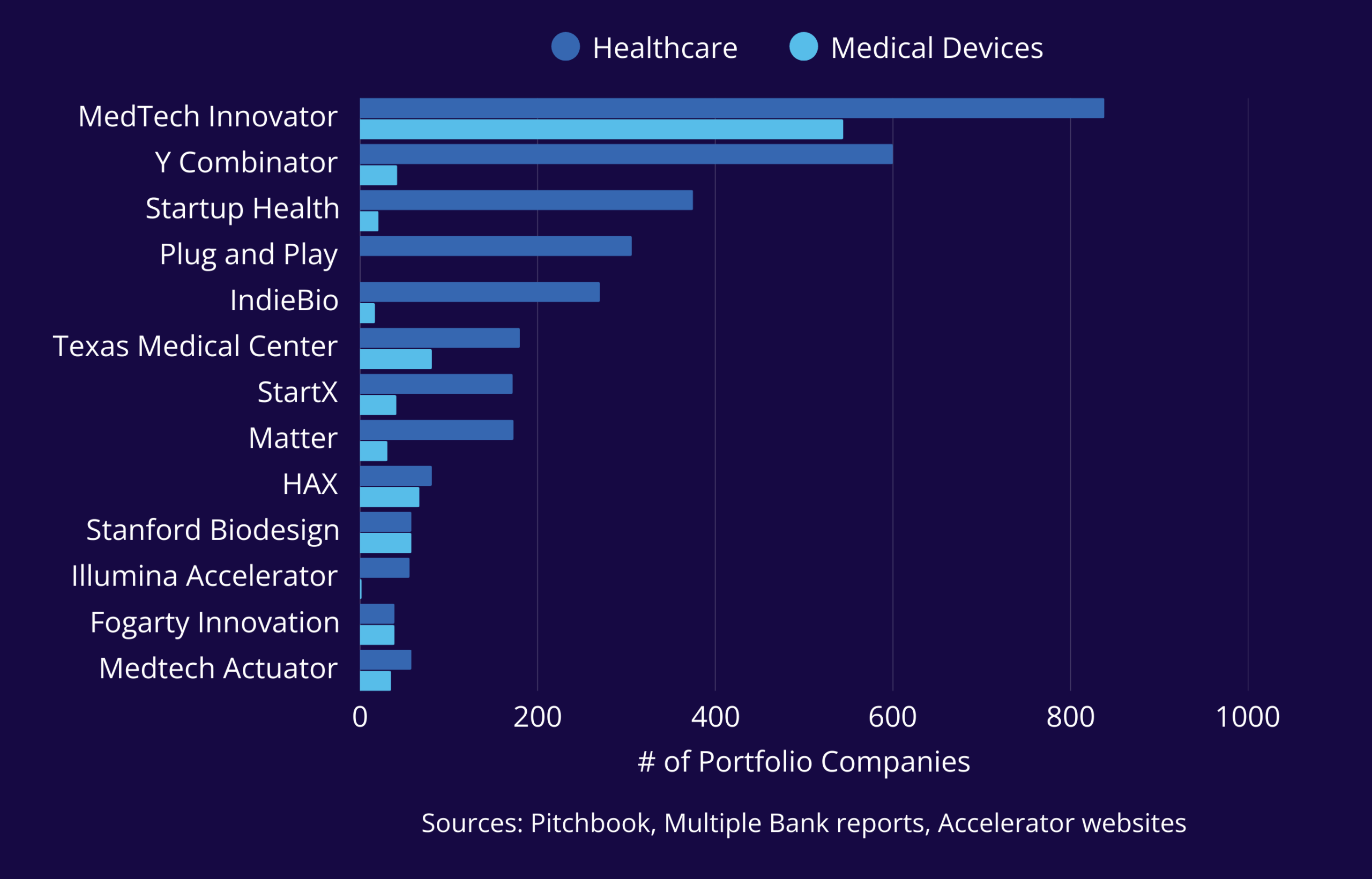
MedTech Innovator is the largest accelerator of Life Science companies in the world. The dominant part of our portfolio is Medical Devices, in addition to our leadership position as an accelerator of diagnostics, digital health, and life science tools technologies. 92% of our portfolio are still in business or have been acquired, and 64% of our companies have raised equity financing following program participation, many within 12 months of participation. In 2025, approximately 40% of total deals in the medtech sector went to MedTech Innovator alumni.
MedTech Innovator US Program Overview

MedTech Innovator awards more than $1 million+ in cash prizes as well as other awards across our US, APAC, and BioTools programs. Each program has its own cohort and includes several rounds where companies gain increasing amounts of exposure to industry leaders including through online application reviews, pitch events, and audience competitions at several major industry conferences. The details below apply primarily to our US program.
MedTech Innovator Radar Forum – Approximately 10% of our 1,800+ applicants are advanced to pitch at the Radar Forum in April, or at our virtual pitch event. During this high-value, engagement and diligence forum, presenters receive feedback from up to 250 judges including investors, clinicians, and senior executives from our leading industry partners such as Johnson & Johnson, Olympus, Edwards Life Sciences, LivaNova, Samsung Research America, Asahi Intecc, Zimmer Biomet, Dexcom, and many others.
MedTech Innovator Accelerator – Based on feedback from the pitch events and subsequent diligence, 50+ of the most innovative companies from around the world are selected to the Accelerator, a 4-month virtual program from June – October. Companies receive coaching and high-profile visibility at the Wilson Sonsini Medical Device conference in June and The MedTech Innovator Showcase program at The MedTech Conference, powered by AdvaMed, in October. Among many benefits, MTI companies will pitch and be featured in expert panels in front of industry and investors at multiple conferences, and they gain access to exclusive partnering and networking opportunities with the CEOs of large medtech companies.
Corporate Mentorship – Our early-stage cohort companies have an unparalleled opportunity to receive continual in-depth, customized mentorship from senior leaders in the medtech industry.
The Accelerator program concludes with all cohort companies participating in the MedTech Innovator Showcase, with the opportunity to compete for a spot presenting in the finals at The MedTech Conference or in the Mid-Stage competition at MedTech Strategist in November.
For those focused on Asia - MedTech Innovator Asia Pacific
MedTech Innovator offers a parallel program and competition providing content regarding accessing and operating in the Asia Pacific region in an Accelerator and US$300K competition in front of industry leaders at the Asia Pacific MedTech Forum in the fall. Companies may apply to both our US-based and Asia programs. Please visit our MedTech Innovator Asia Pacific page for more information on our Asia Pacific program.
For Life Science Tools companies- BioTools Innovator
BioTools Innovator offers the first dedicated accelerator program focused on the unique needs of life science tools companies, with four months of mentorship and culminating with a showcase and $250K competition. Please visit our BioTools Innovator website for more information on our BioTools program.
Meet Our Team

Paul is the founder and CEO of MedTech Innovator. He created and managed MedTech Innovator as a program within RCT Ventures from 2013 – 2016. In 2016, with the financial support of RCT and other sponsors, Paul left to run MedTech Innovator as a stand-alone company. He spent 11 years as Director and Managing Director at RCT Ventures, the investment arm of Research Corporation Technologies, Inc. Prior to RCT, Paul was co-founder and CEO of startups in the medtech, pharma, and tech sectors.

Kathryn Zavala is COO and BioTools Innovator Managing Director at MedTech Innovator. She earned her PhD in Molecular, Cell and Developmental Biology from UCLA. Kathryn has been with MedTech Innovator since 2015. She was a Technology Fellow at the UCLA Office of Intellectual Property, where she assisted with the commercialization of university technologies.

Brian Benson is the Senior Vice President at MedTech Innovator. Prior to joining MedTech Innovator, Brian was the director of entrepreneurship and commercialization at the California NanoSystems Institute (CNSI) where he was responsible for all aspects of the Magnify incubator and driving strategic initiatives for the Institute. He brings over 15 years of experience accelerating the growth and success of transformative life science startups to the team.

Fredrik is the Managing Director Asia Pacific at MedTech Innovator, based in Singapore. He was most recently the Founding CEO of Asia Pacific Medical Technology Association (APACMed). Fredrik brings over 30 years of healthcare industry experience, 25 of which were spent in Asia-Pacific. He has held senior sales/ marketing, general management and strategic consulting positions. Fredrik serves as Board Member and advisor to several healthcare startup ventures and is a frequent speaker on the healthcare industry in Asia-Pacific. He holds a BSc from Gothenburg University, Sweden, and an MBA from Henley Management College, UK.

Daphne Radfar is Chief of Staff at MedTech Innovator. She brings more than 20 years of experience in executive leadership, risk management, and operations in health, education, and startups to this position. Prior to joining MedTech, Daphne served as Chief Operating Officer at Speak UP, working to increase access to quality public education across California. Prior to this role, as Chief Operating Officer of Insure the Uninsured Project, Daphne was instrumental in the organization’s efforts to achieve healthcare reform in California. Daphne received her Bachelor’s in Education, Sociology with an emphasis in Business Administration from UCLA.

Ayelet Marom is the Program Director for BioTools Innovator at MedTech Innovator. She earned her Ph.D. in Cancer Immunology from The Weizmann Institute in Israel, and a Postdoctoral Professional Masters in Bioscience Management from KGI, Claremont alongside her post-doctoral research at City Of Hope. Ayelet is experienced in the innovation ecosystem; prior to MedTech Innovator, she was a senior program manager at Larta Institute, where she managed commercialization programs for science-based Innovations, as well as co-founded and led the product development in a digital health Startup. She is passionate about bringing technology and healthcare closer together to make a real impact on human health.

Nicole is the Program Director for MedTech Innovator. Nicole grew up in Michigan before attending Boston University to study Biomedical Engineering as Trustee Scholar. In 2020, Nicole completed her PhD at Harvard University, working on interdisciplinary medtech projects between the Wyss Institute for Biologically Inspired Engineering and Mass Eye and Ear hospital. After graduating, Nicole launched a startup company, Beacon Bio, based on her PhD work as a Gliklich Healthcare Innovation Scholar. Following Beacon Bio’s acquisition by Desktop Metal, Nicole served as the Vice President of Biofabrication in their Desktop Health division. Nicole’s accolades include the Collegiate Inventors Competition Graduate Winner (2018), the Baxter Young Investigator Award (2020), the Lemelson-MIT Student Prize (2021), and the MIT Technology Review Innovators Under 35 (2023).

Jim is an engineer turned entrepreneur. With a master’s in both Biomedical Engineering Product Development and Entrepreneurship, he blends the business with the technical. Jim previously worked in life sciences and medical devices helping to bring new products to market. He served as Co-Founder and CEO of his most recent company, Clara Biotech, which was founded in 2018 and was acquired by Innovaprep in the summer of 2023.

Glen is Senior Programs Director, Asia Pacific at MedTech Innovator, based in Singapore. He has led successful global programs at Enterprise Singapore’s Singapore Week of Innovation and Technology mega event (SWITCH), helping startups in access the Singapore Market and expand into Asia Pacific. Glen is also the founder of 2 startups and serves as a Venture Partner at Meet Ventures. In his free time, Glen mentors young first time startup founders in Polytechnics and Universities.

Jerry Ciolino is a Director of Operations at MedTech Innovator. He has experience working in multiple accelerator programs with different verticals. He was the Program Manager at Startup UCLA, which caters to undergraduates, graduate students, and recent alumni. More recently, he was the Lead Operations Associate for the Techstars Music Accelerator. He received his degree in Sociology from UCLA.

Council is Program Director, VANGUARD. Prior to joining MedTech Innovator, Council was the clinical research manager for the Institute for Trauma Recovery at UNC-Chapel Hill where she was responsible for managing numerous multi-site clinical trials teams and the Institute’s contracts and grants. She received her BA from UNC-Chapel Hill.

Heather Lukas is the Program Manager for BioTools Innovator VANGUARD, an Enabling Technologies Hub in partnership with BARDA. She recently received her Ph.D. in Medical Engineering from Caltech, studying point-of-care diagnostics and novel techniques for continuous biochemical sensing for wearable device applications. Prior to joining MTI full-time, she was a Senior Fellow with the accelerator, working with MTI cohort companies and leading the team of graduate student fellows in start-up due diligence. She received her B.S. in biomedical engineering with a business minor from Cornell University in 2019.

Eve Jimenez is Marketing Technology Specialist at MedTech Innovator. Prior to joining MedTech Innovator, Eve supported Speak UP’s technology and communications work helping to improve kids’ access to quality education. She received her B.A from Dartmouth College and M.S. from UC Davis

Gabriella Dardano is a Marketing and Events Assistant at MedTech Innovator. She obtained her B.S in Business Administration from Pepperdine University, in Malibu, CA. Prior to joining MedTech Innovator, Gabriella gained substantial experience in event management and marketing at C3 Alliance Corp., a Vancouver-based company in the natural resource sector.

Bernice is the Assistant Manager in Marketing & Events for Asia Pacific at MedTech Innovator, based in Singapore. She is a seasoned Marketing & Events Professional renowned for her exceptional expertise in devising and implementing strategic initiatives that fuel growth. Bernice expertly manages various simultaneous events like networking, hybrid functions, and expos, showcasing her exceptional organizational skills and meticulous attention to detail for flawless coordination across all aspects of these diverse gatherings.

Peter Kemper is Manager of Software Engineering with over 5 years of software development experience in the fintech, energy, and education industries and is based out of San Diego, CA. He graduated from California State University, San Marcos in 2019 with a Bachelor’s in Computer Science.

Nerissa is the Program Manager at Medtech Innovator Asia Pacific, where she fosters the development and growth of medtech startups. Her extensive program management background spans across public, private, and diverse industry sectors, bringing a dynamic range of expertise to her role. Previously, Nerissa led innovation-focused programs at a prominent government agency in Singapore, nurturing the startup ecosystem. Following that, at the Development Bank of Singapore (DBS), she designed and managed financial literacy programs for parents and children.

Jermaine is the Partnership Manager for MedTech Innovator Asia Pacific, supporting strategic collaborations across the region. Before joining MedTech Innovator, she managed international partnerships and innovation programs at Enterprise Singapore, leading initiatives connecting startups, corporates, and government agencies across Asia and North America.

Alicia MacDonald is a first-year MSc student in Biomedical Engineering at UC Irvine, where her thesis research focuses on neurocomputational modeling to identify novel EEG biomarkers for the detection and treatment of epilepsy. She has four years of engineering experience from her time at Medtronic Neurovascular. Alicia earned her B.S. in Mechanical Engineering with a Biomedical Specialization from the University of Alberta in 2021. She is passionate about neuroscience and the next frontiers of neurotechnology.

Devanshi Agarwal is a third-year PhD student at UC San Diego where she is researching the role of rhomboid membrane proteins on alleviating aggregation stress as seen in diseases such as cystic fibrosis, neurodegeneration, and cancer. She received her B.S. from UCLA in 2021 in Biochemistry and Art History, after which she worked in early R&D for a year at Cytokinetics Inc in San Francisco. She is passionate about making the life sciences a more equitable community for everyone.

Jared Cristobal is a Fellow at MedTech Innovator and an MBA candidate at the University of Southern California. He held positions at Poseida Therapeutics and Amgen, where he supported cell and gene therapy manufacturing and process optimization. His experience bridges bioprocessing and business strategy, with a focus on translating scientific innovations into real-world applications. Jared received his BS in Bioengineering from the University of California, Riverside, and his MEng in Biopharmaceutical Processing from Keck Graduate Institute.

Jenna Wittich is a Fellow at MedTech Innovator and a graduate student pursuing an MBA with a focus on healthcare, product leadership, and innovation. She holds an M.S. in Human-Centered Engineering from Tufts University and a B.S. in Engineering and Art History, with a strong foundation in human factors, usability, and systems thinking. Jenna brings experience across product design, product management, and UX research within healthcare and medical technology. Previously, she worked at MITRE as a consultant supporting large-scale healthcare and EHR modernization efforts, collaborating closely with clinicians, patients, engineers, and informaticists to improve usability, safety, and care delivery.

Sachi Bansal is a Bioengineering Ph.D. candidate in the Anatomical Engineering Group at UCLA. Her research focuses on the development and surgical integration of novel prosthetic devices. Sachi received her B.S. in Biology from the University of Washington Seattle in 2019 after which she worked first for a CRO managing clinical trials and then at Stanford conducting clinical research on the long-term effects of ACL tears.

Sarah Fatkin is a third-year PhD candidate in the Pyle Lab at UCLA, where she engineers human stem cell models to study limb formation during early development. Prior to graduate school, she worked in the Frye Lab at UCLA investigating the neurobiology of perception using virtual reality. She received her B.S. in Neuroscience and B.A. in European Languages from UCLA in 2022.

Shelby Guerena is a Fellow at MedTech Innovator and an MBA candidate at UCLA Anderson School of Management, where she specializes in Healthcare Management. She brings deep experience in healthcare technology and strategy from Deloitte Consulting, where she was an early member of the Smart Hospital practice, leading clinical command center implementations, defining digital health strategies, and driving enterprise data initiatives for major health systems and life sciences organizations. She holds a B.S. in Business Administration with a minor in Biology from the University of Colorado, Boulder. Shelby combines consulting rigor, healthcare domain expertise, and venture insight to support the growth of innovative healthcare companies.

Sophia Cao is a first-year MBA student at UCLA Anderson School of Management. She brings financial expertise and industry knowledge from her experience as a Life Sciences Banking Associate at J.P. Morgan, where she supported post-Series A through publicly held companies across Southern California. Sophia received her B.A. in Economics with a minor in Biological Sciences from the University of Chicago in 2022. She is passionate about applying finance to help scale innovation and drive long-term value creation.
Meet Our Board

Scott Whitaker is President and CEO of AdvaMed, the world’s largest medical technology association. He is widely recognized as a top health care advocate and policy expert with experience across multiple health policy sectors, having worked successfully with diverse stakeholder groups – including hospital, patient and physician groups, regulators, public and private payers, and global trade organizations – to advance medical technology innovation and timely patient access to next-generation treatments and cures.

Chris Martin is the President of RCT Ventures, the investment arm of Research Corporation Technologies, which is focused on investing in early-stage biomedical companies.

Biren Mehta is the Vice President, Venture Investments – MedTech at Johnson & Johnson Innovation. He is responsible for existing and new MedTech investments on the West Coast, ensuring targeted strategic options are shaped for success. Biren plays a critical role in building and maintaining networks in the venture capital community, entrepreneurs, startups, incubators and accelerators and other venture organizations that fit the strategic interests of the MedTech sector, sourcing new opportunities for the sector and positioning JJDC as the partner of choice.

Karen is an independent board director and healthcare executive with 35+ years of experience in highly regulated insurance lines of business, product development, direct to consumer growth, compliance and organizational transformation. She currently serves as CEO of Clever Care Health Plan. Formerly Medicare Officer at Health Net, a Centene Corporation company, she led a $3.5B+ business and previously held executive roles at UnitedHealthcare, Cigna, WellCare, Health Essentials, Cleveland Clinic Health System and Lake Health. She advises healthcare tech firms and champions leadership development and health equity.
