Neural Analytics Announces First Site Initiation, First Subject Enrollment in the CODEX Study
Neural Analytics Inc., a medical robotics company developing and commercializing technologies to measure and track brain health, has announced the first site initiation and first subjects enrolled into the CODEX Study, a prospective, single arm multi-center safety and technical feasibility study utilizing an investigational iteration of the Lucid Robotic System (Lucid M1 Transcranial Doppler Ultrasound System and NeuralBot System), at the Ochsner Clinical Foundation in New Orleans, La.
“There is a growing interest in the diagnostic potential of measuring the dynamic changes of cerebral blood flow in different neurological conditions. Participating in the CODEX Study using the Lucid Robotic System to offer an autonomous measurement of cerebral blood flow velocities has been exciting. We look forward to advancing the field and expanding the clinical relevance of non-invasive measurement of cerebral blood flow in neurological conditions,” said Ifeanyi Iwuchukwu, M.D., assistant professor of Neurocritical Care and Stroke at The Ochsner Clinical Foundation.
Dr. Iwuchukwu and the Ochsner Clinical Foundation are the first to enroll subjects into the global multi-center study. Although the Lucid Robotic System is a CE-Marked and U.S. Food and Drug Administration-cleared device, the primary objective of the CODEX Study is to employ an investigational iteration of the Lucid Robotic System to gather and further explore the technical feasibility of the technology in a wider range of patients and neurological pathologies where the monitoring of cerebral blood flow velocities (CBFV) would be of benefit.
“We are pleased and privileged that we are able to partner with Dr. Iwuchukwu and the Ochsner Clinical Foundation in the enrollment of our first patients into the CODEX Study,” said Robert Hamilton, Ph.D., co-founder and chief science officer of Neural Analytics. “This is a big milestone for us, as we are committed to advancing brain healthcare through transformative technology that empowers clinicians with the critical information needed to make clinical decisions and improve patient outcomes.”
Each year, neurological diseases cost the U.S. healthcare system nearly $800 billion.2 Traumatic brain injury, migraine, Alzheimer’s and stroke account for more than $438 billion of that cost.1 Stroke affects more than 795,000 people in the United States— someone has a stroke every 40 seconds, and someone dies from stroke every four minutes.2 It is a time-sensitive disease and requires intervention within 24 hours of onset of symptoms.3 Incorrect assessment of large vessel stroke leads to misdiagnosis and treatment delays, resulting in death or disability for stroke patients. Despite recent advances in life-saving treatments for acute ischemic stroke, less than five percent of stroke patients qualify for intervention because they do not present early enough.4 The American Heart Association/American Stroke Association Guidelines for Early Management of Acute Ischemic Stroke state that detection of large vessel occlusion by means of noninvasive intracranial vascular imaging greatly improves the ability to make appropriate clinical decisions.5
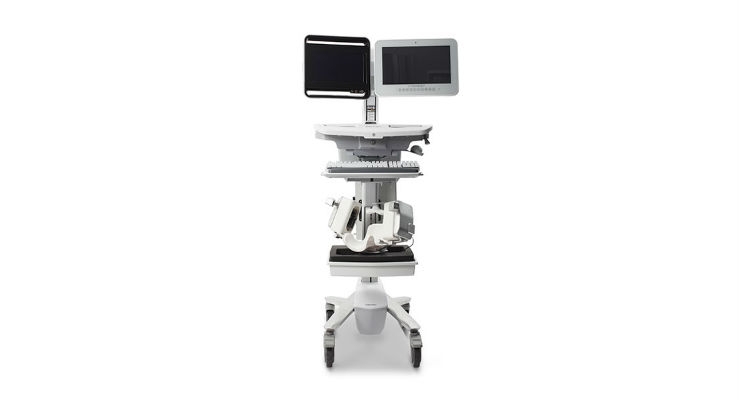
The Lucid Robotic System is a medical ultrasound device intended for use as an adjunct to standard clinical practices for measuring and displaying CBFV and the occurrence of transient emboli within the brain. The device is not intended to replace other means of evaluating vital patient physiological processes. Neural Analytics is actively enrolling patients into this research feasibility study and intends on activating up to 15 sites, both domestically and globally.
Ochsner Health System is Louisiana’s largest non-profit, academic, healthcare system. Driven by a mission to Serve, Heal, Lead, Educate and Innovate, coordinated clinical and hospital patient care is provided across the region by Ochsner’s 40 owned, managed and affiliated hospitals and specialty hospitals, and more than 100 health centers and urgent care centers. Ochsner is the only Louisiana hospital recognized by U.S. News & World Report as a “Best Hospital” across three specialty categories caring for patients from all 50 states and more than 70 countries worldwide each year. Ochsner employs nearly 25,000 employees and over 4,500 employed and affiliated physicians in over 90 medical specialties and subspecialties and conducts more than 700 clinical research studies.
Neural Analytics Inc. is a medical robotics company developing and commercializing technologies to measure and track brain health. The company’s Lucid Robotic System (Lucid M1 Transcranial Doppler Ultrasound System and NeuralBot System) is a robotically assisted ultrasound system for brain health assessment. It combines an all-in-one neurovascular ultrasound device, designed to non-invasively measure and display brain blood flow information under the guidance of a healthcare professional. The company’s technology integrates ultrasound, robotics and machine learning to empower neurologists with critical information about brain health to make clinical decisions and improve patient outcomes.

